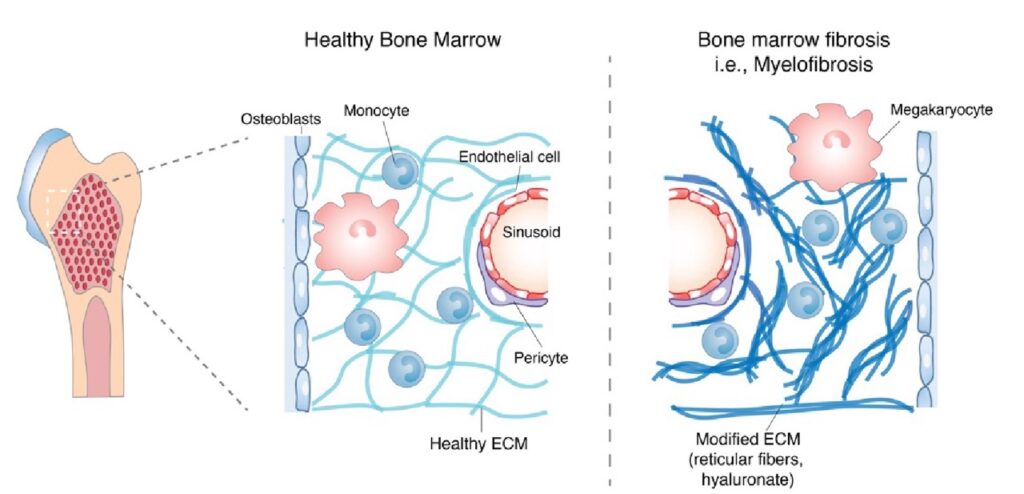
Fibrosis and remodeling of extracellular matrix (ECM) are involved in many diseases affecting health, such as tumors, wound healing, and chronic inflammation. During fibrosis, tissues undergo changes in their viscoelastic properties, i.e., how they resist deformation like a solid and dissipate stress over time like a fluid. Independent of stiffness, an applied stress relaxes rapidly in a more viscous, liquid-like matrix, whereas in a more elastic, solid-like material, stress relaxes slowly. The Vining Lab investigates the impact of viscoelasticity on inflammation in fibrotic tissues and develops new immunotherapies in cancer.
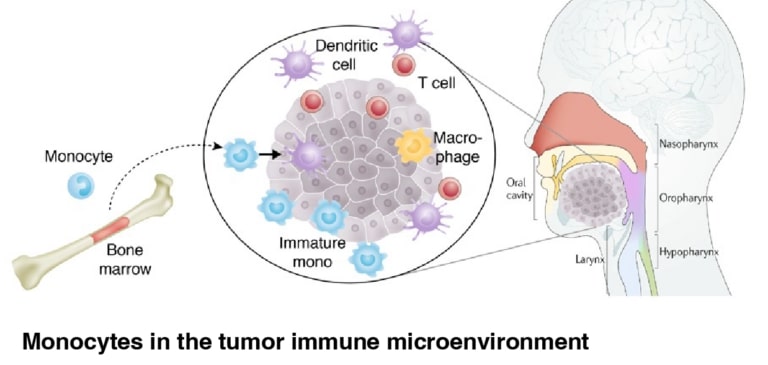
- We utilize an artificial ECM to dissect a mechanical checkpoint of myeloid cell fate and develop strategies to control myeloid inflammation in vivo. Monocytes are a type of white blood cell that impacts the local immune response by producing cytokines and differentiating into phagocytic and antigen-presenting cells, like macrophages and dendritic cells.
- We first investigated this concept in hematopoietic malignancies that result in myelofibrosis, revealing that changes in viscoelasticity regulate inflammation and differentiation of monocytes, and is targetable by a PI3K-gamma inhibitor.
- This work is supported by the NIH/NIGMS and the American Cancer Society, and has broad applications in fibrotic and inflammatory diseases.